Penile Filler Injection Technique: The Fascial Plane Precision Guide
Introduction: Why Technique Is the Only Variable That Matters
The outcome of a penile filler procedure is not primarily determined by the filler product selected. It is determined by the millimeter-level precision of where that product is placed and how it is delivered.
This article addresses a specific reader: the high-information professional who has already researched this topic, understands that marketing language is not a substitute for clinical specificity, and wants to comprehend the actual mechanics before making a decision. For men who never imagined a viable solution existed for their concerns, understanding the technical foundation transforms uncertainty into informed confidence.
This guide explains the exact anatomical planes, device selection logic, entry point strategy, and vascular risk hierarchy in plain language. No medical degree is required to follow along.
The central thesis is straightforward: the sub-Dartos/Buck’s fascial plane is not a preference or a style. It is the only anatomically defensible injection zone, and every deviation from it produces a predictable, measurable category of harm.
A transparency note is warranted upfront: all penile filler procedures use FDA-approved dermal fillers in an off-label capacity. No filler has been specifically FDA-evaluated for penile enlargement indications. This regulatory context makes provider selection and technique expertise the primary safety variables.
While competitor content focuses on outcomes and reassurance, and academic literature targets clinicians, this article bridges that gap for the technically sophisticated patient.
The Anatomy to Understand Before Anything Else
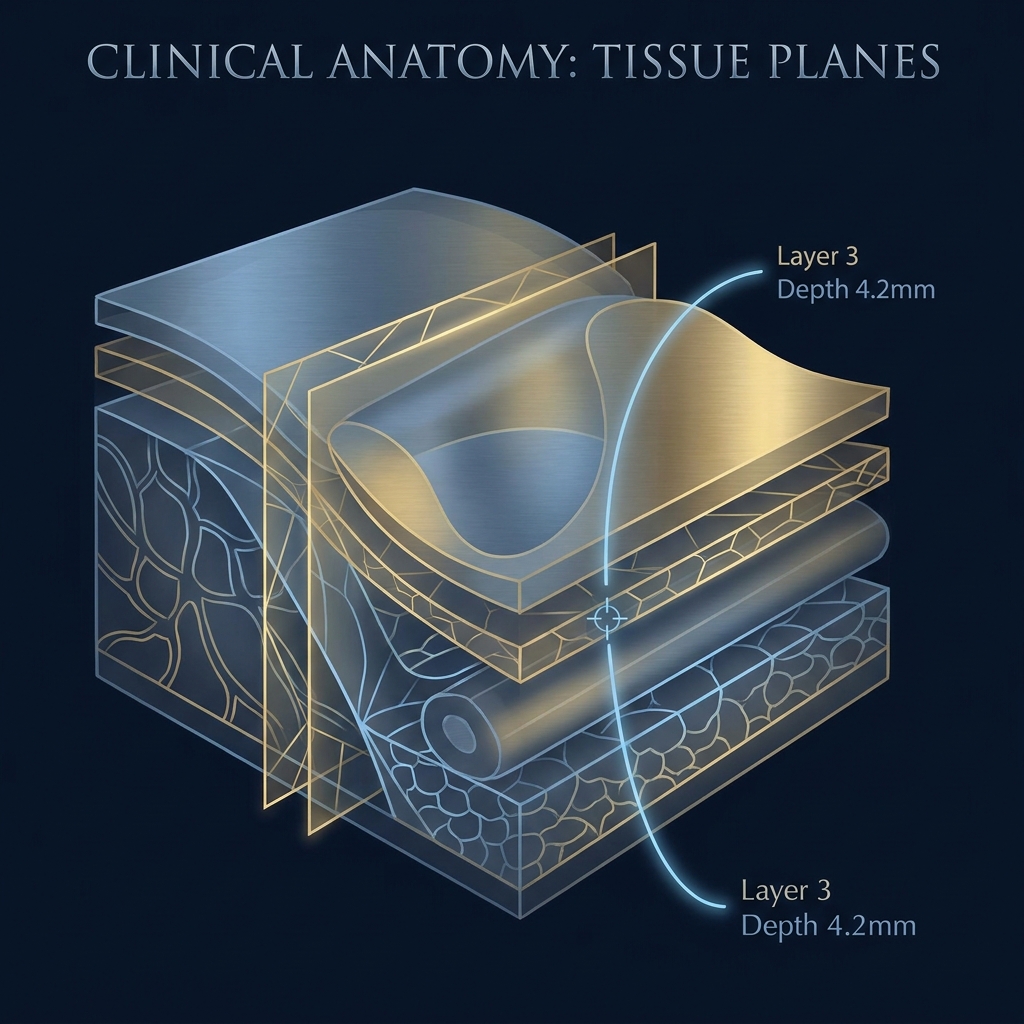
The penis has a layered architecture that proceeds from outside in: skin, dartos fascia (superficial), sub-Dartos space, Buck’s fascia (deep), tunica albuginea, and finally the corpora cavernosa.
The dartos fascia is a thin, mobile, fibromuscular layer directly beneath the penile skin. It functions similarly to the superficial musculoaponeurotic system (SMAS) in facial anatomy.
Buck’s fascia is the dense, inelastic deep penile fascia that encases the corpora cavernosa and corpus spongiosum. Critically, this layer houses the dorsal neurovascular bundle, which includes the dorsal artery, dorsal vein, and dorsal nerve.
The target injection zone is the sub-Dartos space: the potential space between the underside of the dartos fascia and the outer surface of Buck’s fascia. Clinical literature describes this plane as “relatively avascular and easy to detach,” making it the safest corridor for filler deposition.
Filler placed correctly in the sub-Dartos/Buck’s plane does not enter the corpora cavernosa, does not interfere with the erectile mechanism, and does not affect the ability to achieve or maintain erection.
Penile anatomy exhibits significant inter-individual variation. Shaft length, skin mobility, and fascial thickness vary considerably between patients. This variability is precisely why a one-size-fits-all injection protocol is clinically inappropriate.
The glans represents a distinct anatomical zone with higher vascularity, denser lymphatic drainage, and different filler requirements. Shaft girth enhancement achieves satisfaction rates above 80%, while glans injection satisfaction hovers around 50%.
The Only Acceptable Injection Plane and What Happens When It Is Missed
The sub-Dartos/Buck’s fascial plane is the only acceptable injection zone for penile girth enhancement. This is not a marketing claim. It is the consensus position across multi-center randomized controlled trial literature, the CDS technique case report, and peer-reviewed clinical studies.
When filler is placed too superficially (above the dartos fascia, in the subcutaneous tissue), the consequences include visible surface irregularities, palpable nodularity, the Tyndall effect (a bluish discoloration caused by light scattering through superficially placed hyaluronic acid), and filler migration along tissue planes.
When filler is placed too deeply (at or below Buck’s fascia), proximity to the dorsal neurovascular bundle creates risk of vascular compression, occlusion, or direct nerve trauma.
The sub-Dartos space is a narrow anatomical corridor. Precise depth control is not a refinement; it is the foundational requirement. A 2025 Cureus case report introducing the CDS technique explicitly states the procedure “should be reserved for experienced practitioners with specific training in male genital anatomy.”
The connection between depth error and complication type is direct: superficial placement leads to aesthetic failure (nodules, Tyndall effect, migration), while deep placement creates functional and vascular risk (occlusion, nerve compression). Each error category has a distinct clinical signature and management pathway.
Needle vs. Cannula: The Device Selection Decision Explained
Two primary injection devices exist: sharp-tip needles and blunt-tip microcannulas. Each serves a distinct role in the procedure workflow.
The sharp needle is used to create the initial entry point, puncturing through skin and dartos fascia. Its role is access, not distribution.
The blunt-tip cannula is introduced into the sub-Dartos plane once the entry point is created, then advanced to distribute filler across the shaft. The blunt tip deflects around vessels rather than piercing them, reducing (but not eliminating) vascular injury risk.
A 2025 ScienceDirect narrative review found that cannulas are associated with lower bruising rates and reduced vascular occlusion signal in vessel-dense regions. However, arterial wall penetration and ischemic events remain possible with improper plane selection or excessive injection pressure.
Cannulas are not inherently risk-free. The cannula’s safety advantage is mechanical (blunt deflection), not absolute. If the operator selects the wrong plane, injects at high pressure, or delivers a large bolus volume, the risk profile escalates regardless of device type.
Gauge selection affects tissue trauma, flow rate, and control. The CDS technique uses an 18-gauge blunt-tip cannula through a single mid-shaft entry point. Multi-center RCT protocols have used 22-gauge cannulas at four entry sites. The principle remains consistent: sharp needle for access, blunt cannula for distribution.
Entry Point Strategy: Where the Cannula Goes In and Why It Matters
Entry point selection determines the distribution geometry of the filler and how evenly it can be spread circumferentially and longitudinally across the shaft.
A multi-center RCT protocol used four entry sites at the 2 o’clock and 10 o’clock positions at both the penile base and distal shaft. This quadrant approach allows the cannula to reach all shaft zones from a limited number of punctures.
The single-entry CDS technique uses one mid-shaft entry point, with the 18-gauge cannula advanced proximally and distally to deposit filler in structured micro-droplets across the entire shaft. This approach minimizes puncture trauma but requires higher operator skill to achieve circumferential symmetry from a single corridor.
Entry points are selected at dorsolateral positions (2 o’clock and 10 o’clock) to avoid the midline dorsal vein and the ventral urethra. The dorsal midline (12 o’clock) is avoided because it overlies the superficial dorsal vein and deep dorsal artery. The ventral midline (6 o’clock) is avoided because it overlies the corpus spongiosum and urethra. Lateral and dorsolateral positions represent the lowest vascular risk corridors.
Filler Distribution Techniques: Threading, Fanning, and the CDS Micro-Droplet Method
Three primary distribution techniques are used in penile filler injection: linear threading, fanning, and the structured micro-droplet approach.
Linear threading involves advancing the cannula to the distal extent of the target zone, then depositing filler in a continuous column as the cannula is withdrawn. The risk includes uneven column distribution and palpable ridging if volume is not precisely controlled.
Fanning redirects the cannula at multiple angles from the same entry point, depositing filler in a fan-shaped distribution. While this provides broader coverage from fewer punctures, overlapping deposits at the pivot point can create focal nodularity.
The CDS micro-droplet method deposits filler in structured small aliquots at controlled intervals rather than as a continuous column or fan. This approach produces more even volumetric distribution and reduces risk of focal accumulation. It requires precise pressure control and consistent aliquot sizing.
Traditional serial puncture needle injection is associated with the highest complication rate. Multiple sharp-needle passes increase vascular trauma risk, create multiple potential migration pathways, and produce less even distribution than cannula-based techniques.
Post-procedure massage for approximately one week after the procedure is a clinical tool used to facilitate even filler distribution and correct minor asymmetries before the product fully integrates. Understanding the full penile dermal filler procedure timeline helps patients set accurate expectations for each phase of recovery.
Filler Selection: Why Hyaluronic Acid Is the Starting Point for First-Time Patients
The filler landscape includes hyaluronic acid (HA), polylactic acid (PLA), polymethylmethacrylate (PMMA), silicone, and non-medical self-injected materials. Each carries a distinct risk and reversibility profile.
HA is the clinical consensus starting point. A 2023 systematic review and meta-analysis found that HA increased penile diameter more and achieved better patient satisfaction than PLA. Standardized low-volume HA protocols have the lowest complication rates across all filler categories.
The reversibility advantage of HA is critical: hyaluronidase dissolves HA within hours, providing an emergency reversal mechanism that no other volumizing material can match. This reversibility is the safety backstop that makes HA the only appropriate choice for first-time patients.
HA results typically last 12 to 24 months, with some protocols reporting longer duration with maintenance sessions. Approximately 6 to 8 mL of filler produces roughly 0.5 inches of circumferential girth increase, though the exact ratio depends on baseline anatomy and tissue elasticity.
Major aesthetic medicine organizations, including the Sexual Medicine Society of North America and the European Association of Urology, have released statements advocating for avoidance of permanent fillers in penile augmentation. When complications occur with non-dissolvable materials, management requires wide surgical excision down to Buck’s fascia.
Vascular Risk Hierarchy: The Anatomy of What Can Go Wrong
The vascular risk hierarchy of the penile shaft places the dorsal neurovascular bundle (running at the 12 o’clock position under Buck’s fascia) as the highest-risk structure. The circumflex veins at lateral positions represent intermediate risk. The corpus spongiosum and urethra at the 6 o’clock position constitute a distinct risk category.
Two mechanisms cause vascular injury: extravascular compression (filler volume compresses an adjacent vessel from outside) and intravascular embolism (filler is injected directly into a vessel, blocking flow distally).
Risk factors for intravascular occlusion include high injection pressure, large volume bolus injections, use of small sharp needles in vessel-dense regions, and deeper injection planes. These factors underscore why cannula-based, low-pressure, multi-session protocols are preferred.
A 2025 PMC review describes five clinical stages of ischemia post-filler injection: Stage I (blanching), Stage II (livedoid changes), Stage III (skin barrier deterioration at approximately 72 hours), Stage IV (coagulative necrosis at 5 to 10 days), and Stage V (eschar formation).
Early hyaluronidase administration, ideally within 48 hours of vascular occlusion, is associated with significantly better outcomes. Hyaluronidase can traverse blood vessel walls and dissolve intravascular HA, making it the critical emergency reversal agent.
Data presented at the 2024 American Urological Association meeting documented nearly 500 patients treated with HA filler, showing complication rates under 2% with no serious adverse events, no reported erectile dysfunction, and no loss of sensitivity. This penile filler procedure safety record reflects the outcomes achievable when technique and provider selection standards are rigorously applied.
The Role of Ultrasound Guidance in Plane Confirmation and Vascular Avoidance
Ultrasound guidance adds real-time visualization of the needle or cannula tip position relative to the dartos fascia, Buck’s fascia, and adjacent vascular structures. This enables confirmation that the device is in the correct anatomical plane before filler is deposited.
Tactile feedback (the “give” felt when entering the sub-Dartos space) is a legitimate technique cue, but it is operator-dependent and cannot visualize vascular structures. Ultrasound adds an objective, real-time safety layer.
Ultrasound guidance is increasingly used in advanced protocols but remains absent from most clinic-level competitor content, indicating it is a differentiating capability rather than a universal standard.
Asking a prospective provider whether they use ultrasound guidance, and why or why not, is a technically informed question that reveals the depth of their anatomical training and procedural sophistication.
Physician Training: What “Qualified Provider” Actually Means in This Context
Physician training requirements for penile filler injection are not formally standardized at the national level. “Trained provider” language used by most competitor content is vague and does not specify what training actually entails.
Relevant training includes licensed physician status (MD or DO), specialization in sexual medicine, urology, or a closely related field with advanced knowledge of male genital anatomy, specific training in advanced filler injection techniques, and familiarity with vascular complication recognition and hyaluronidase emergency protocols.
American Board of Urology certification requires completion of a minimum 5-year ACGME-accredited urology residency (12 months general surgery, 36 months clinical urology), passage of written qualifying and certifying exams including pathology and uroradiology. This credential establishes the anatomical foundation but is the starting point, not the complete qualification.
Leading protocols require providers to be licensed physicians specializing in sexual medicine or urology with specific training in male genital anatomy and advanced filler techniques.
Stoller Medical Group, with over 15,000 enlargement procedures performed, exemplifies the volume of experience that builds technique mastery. Dr. Roy B. Stoller’s 25 years of experience in aesthetic and restorative medicine, including 5 years dedicated specifically to non-surgical male enhancement, represents the depth of specialization that technically sophisticated patients should seek.
Contraindications and Patient Selection: Who Should Not Have This Procedure
Primary contraindications include uncircumcised status (for shaft enhancement), active infection at or near the injection site, bleeding disorders or anticoagulant therapy, unrealistic expectations about outcome magnitude, and significant buried penis anatomy.
The uncircumcised contraindication has anatomical basis: the foreskin creates a mobile tissue envelope that can trap migrating filler, restrict retraction as filler volume increases, and produce asymmetric contouring. This is not a preference; it is an anatomical incompatibility with safe shaft enhancement.
The Sexual Medicine Society of North America and the European Association of Urology both advocate for psychological evaluation as part of patient selection, recognizing that body dysmorphic disorder and unrealistic expectations are meaningful risk factors for poor outcomes regardless of technical execution.
Leading protocols use multiple sessions rather than single large-volume injections. Staged treatment reduces per-session vascular load, allows healing interval assessment, improves symmetry, and enables incremental volume calibration to individual anatomy. This staged treatment approach is standard protocol at Stoller Medical Group.
Post-Procedure Protocol: What Happens After the Injection and Why Each Step Is Clinical
The standard post-procedure protocol follows a clinical sequence: compression bandage for approximately one day (reduces hematoma formation and supports initial filler positioning), antibiotics and NSAIDs for approximately three days (infection prophylaxis and inflammation management), sexual activity abstinence for one to four weeks (prevents mechanical displacement of filler before integration), and daily massage for approximately one week (facilitates even distribution and corrects minor asymmetries).
Post-procedure massage is not cosmetic aftercare. It is a technique for redistributing filler micro-deposits before the product fully integrates with surrounding tissue. The window for massage-based correction closes as HA hydrates and stabilizes, typically within the first week.
The healing interval between staged sessions is typically two to three months. This interval allows complete tissue integration, accurate assessment of distributed volume, and identification of any asymmetry or nodularity requiring correction.
Recovery timeline is practical: patients return to normal daily activity within approximately 10 days, with sexual activity resumable within 7 to 10 days. A detailed overview of non-surgical penile enhancement recovery outlines what to expect at each stage following the procedure.
Patients should monitor for any blanching, mottling, or color change at the injection site in the first 48 to 72 hours. These are potential early ischemia signals requiring immediate provider contact, not symptoms to observe and wait on.
Conclusion: Technique Is the Standard, Not the Selling Point
The outcome of a penile filler procedure is determined by the millimeter-level precision of anatomical plane selection, the device and technique used to deposit the product, the provider’s vascular anatomy training, and the protocol’s ability to recognize and respond to early complications.
The measurable standards a technically sophisticated patient should evaluate include: correct injection plane (sub-Dartos/Buck’s fascial space), cannula-based distribution technique, entry point selection that avoids the dorsal neurovascular bundle and ventral urethra, HA as the starting filler for its reversibility advantage, staged multi-session protocol, and provider training that includes vascular complication recognition and hyaluronidase emergency protocols.
A provider’s ability to recognize Stage I ischemia and intervene within the critical 48-hour window is the single most consequential measure of clinical competence.
Understanding the anatomy, the technique hierarchy, and the vascular risk framework does not make someone a clinician. It does make them a better-informed patient, capable of evaluating providers on the basis of measurable clinical standards rather than marketing language.
Ready to Evaluate Options with Confidence? Start with a Consultation.
For readers who now understand the technical standards, the consultation is the natural next step. It is not a sales conversation; it is the opportunity to apply the evaluation framework this article has provided.
Stoller Medical Group brings relevant credentials to this conversation: over 15,000 procedures performed, a physician-led protocol with advanced training in male genital anatomy, an HA-based staged treatment approach, and multi-location accessibility.
The free consultation offers a low-barrier entry point to ask the technically informed questions this article has equipped readers to ask: questions about injection plane, cannula technique, entry point strategy, hyaluronidase availability, and provider training.
Five locations serve the Northeast and Midwest: Manhattan (515 Madison Avenue), Long Island (Jericho), Albany (Latham), Pennsylvania (Chadds Ford), and Minnesota (Eagan).
The invitation is straightforward: bring the same level of analytical rigor to the provider selection conversation that has been applied to understanding the procedure itself.